In the late 1970s, capnography was developed as a monitoring tool for use during anaesthesia and intensive care following extensive work by Smallhout and Kalenda highlighting its benefits (Gravenstein et al, 1989). Its use spread over time to emergency departments and prehospital care as an adjunct to verify intubation success (Donald and Paterson, 2006).
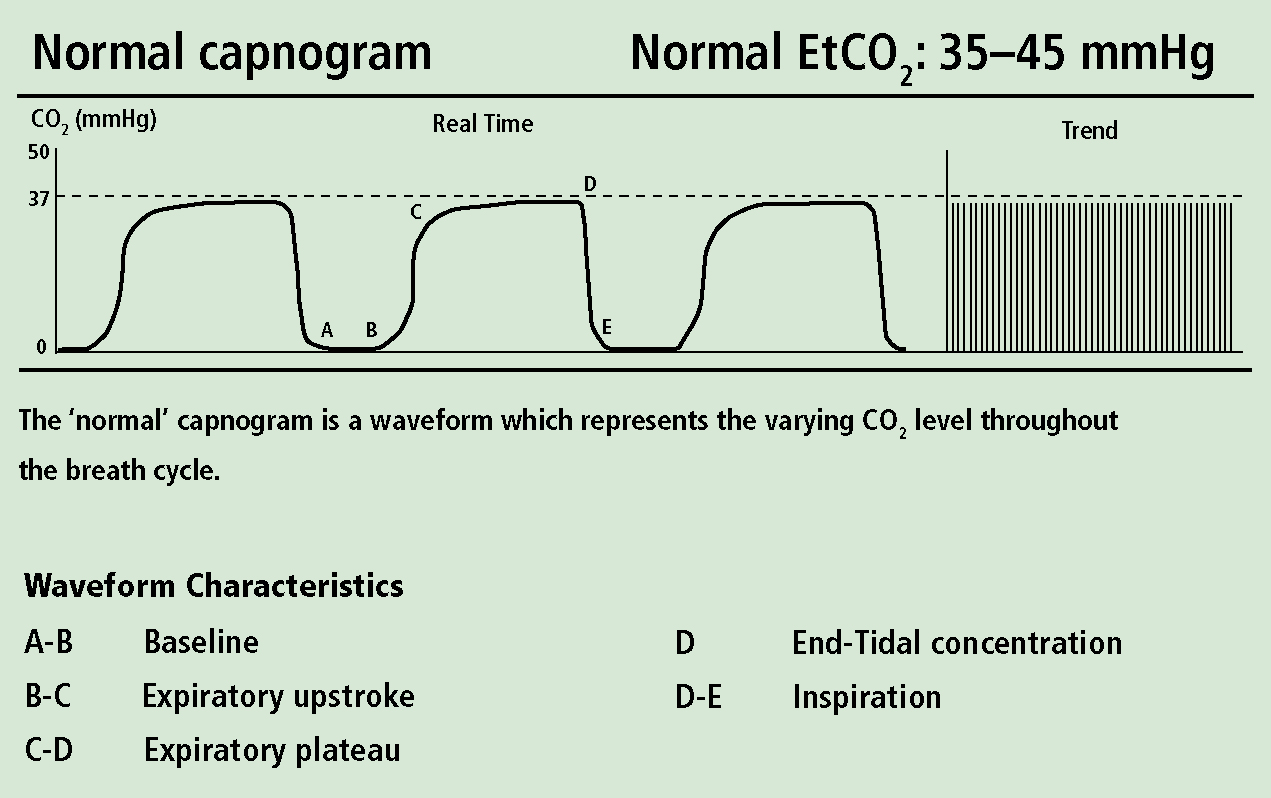
A non-invasive technique, prehospital capnography measures exhaled carbon dioxide through a ‘sidestream’ capnometer; a preset volume of gas is aspirated from the patients expired breath and pumped from an adaptor (positioned on an endotracheal tube or near the patient's airway) to a monitor for analysis (Gilboy and Hawkins, 2006), and accordingly produces a capnogram waveform of expired carbon dioxide against time (Figure 1) (Zwerneman, 2006). Table 1 illustrates some commonly used terms in the study of capnography (Bhende and LaCovey, 2001).
| Term | Definition |
|---|---|
| Capnography | Measurement of CO2 in exhaled breath |
| Capnometer | Numeric measurement of CO2 |
| Capnogram | Waveform produced by monitoring CO2 |
| ETCO2 (or PETCO2) | The concentration (or partial pressure) of CO2 released at the end of expiration |
| PaCO2 | Partial pressure of CO2 in arterial blood. |
An important perception of capnography is its distinction from the more commonly observed SpO2 (saturation percentage of oxygen) which exclusively measures tissue oxygenation (Redford et al, 2004). End-tidal CO2 monitoring is concerned with directly measuring ventilation of the lungs (through CO2 elimination), but additionally is an indirect marker of gauging metabolism (reflecting CO2 production from respiring tissues) and circulation (through transport of waste CO2 to the lungs) (Donald and Paterson, 2006).
An essential understanding in capnography is that ETCO2 is not a measurement of CO2 concentration in the circulation, but an indicator of it—arterial partial pressure of CO2 (PaCO2) is obtained by analysing arterial blood gases, an invasive and expensive procedure requiring complex analysis by complex machinery, currently limited to the hospital setting and only when absolutely necessary (Santos et al, 1994).
Literature review
In order to identify best evidence-based practice for capnography applications, while simultaneously exploring other potential scenarios for use in prehospital care, a literature review was carried out. This encompassed a range of sources to collate current evidence and opinion on the topic. Initially, an online literature search was undertaken, sourcing articles from Medline, PubMed, CINAHL and the Cochrane Library. The ‘PICO’ search strategy was used (which incorporates the headings Population, Intervention, Comparison and Outcome) (Richardson et al, 1995) to retrieve relevant papers around the topic. This template has been shown to increase precision and relevancy of results when compared to standard field searching in evidence-based medicine (Schardt et al, 2007). Both hospital and prehospital based articles were included, as it was felt that some hospital-based studies were decidedly relevant involving scenarios which are readily reproducible in the prehospital setting. Article age was limited to publication years 1988 to 2011, as some pertinent early articles on the topic were appropriate for examining the heredity of capnography. Obtained results were included if relevant to the topic, segregated into their key themes and then evaluated in a structured format.
Intubation
Initially, the primary role for capnography in prehospital care was as an aid to endotracheal intubation; unrecognized oesophageal intubation causes rapidly fatal hypoxia following misplaced endotracheal tube (ETT). Review of the literature is decidedly conflicting and inconclusive over the actual incidence, but rates as high as 17-25% have been reported in prehospital procedures (Falk and Sayne 1999; Lockey and Davies, 2001; Omert et al, 2001; Grmec and Malley, 2004). A metaanalysis by Lah and Grmecl in 2010 concluded that ‘misplaced oesophageal intubations (were) reported….usually by inadequately trained personnel,’ however, no mention is given to their judgement on an operator's education or appropriate experience, therefore conclusions cannot be drawn around suitable training.
A number of methods exist for determining correct ETT placement (lateral/epigastric auscultation, bilateral hemi-thorax expansion, direct laryngoscopy of vocal cords, ventilator compliance, capnography) (Donald and Paterson, 2006), but no single technique has conclusively demonstrated specificity (Grmec and Malley, 2004). Although the efficacy of capnography in determining ETT placement is heavily debated in the literature, a meta-analysis of 2192 cases by Li (2001) established 93% sensitivity and 97% specificity when using solely ETCO2 to confirm placement.
Extrapolating these figures, it could therefore be proposed that 1 in 10 patients may be inappropriately managed as a result of solely using capnography. Recognizing this, the International Liaison Committee on Resuscitation (ILCOR) (2005) highlight the benefits and requirement for capnography but under the precept that the device is not conclusive and an additional confirmation method should be used for ETT placement, a concept sustained in recent guidelines by the American Heart Association (2010) and the UK Resuscitation Council (2010).
Cardiac arrest
Capnography during cardiac arrest has been shown to have an implicit benefit outside of confirming intubation success; as early as 1978, Kalenda et al reported a direct agreement between cardiac output (spontaneous or cardiac compression-induced) and capnography trace (as ETCO2 indirectly reflects pulmonary blood circulation) (Miner et al 2002, Gilboy and Hawkins 2006), which has been confirmed in a number of both experimental and clinical studies, with many demonstrating a linear relationship between cardiac output and ETCO2 (Gudipati 1988, Grmec 2004, Deakin et al 2004).
A retrospective case control study by Pokorna et al (2010) aimed to analyse whether an increase in ETCO2 during cardiac arrest was a reliable indicator of return of spontaneous circulation (ROSC); 140 advanced life support cases were examined (only 108 were included in the results due to lack of ETCO2 in some patients, an anomaly which was not discussed) and divided into groups of ROSC or no ROSC.
Good internal validity was achieved by constant automatic ventilation being a control in the study as any unrestrained alveolar ventilation (i.e. via a bag-valve-mask) would have a marked effect on ETCO2 levels, serving to reduce bias in the non-blinded design. It was found that an increase in ETCO2 of >10 mmHg could reliably indicate ROSC, although the small sample size requires scrutiny. The study does possess significant transferability as all ALS cases were conducted in the prehospital environment, and supports evidence from previous studies.
The converse to this scenario has also been investigated—Cantineau et al (1996), Levine et al (1997), Ahrens et al (2001) and Grmec et al (2001) all found that a sudden decrease in ETCO2 demonstrated a loss of spontaneous circulation, and also that a low initial ETCO2 (<10 mmHg) in cardiac arrest resulted in 0% survival rate (Salen et al, 2001, Grmec et al, 2007).
Bronchospasm
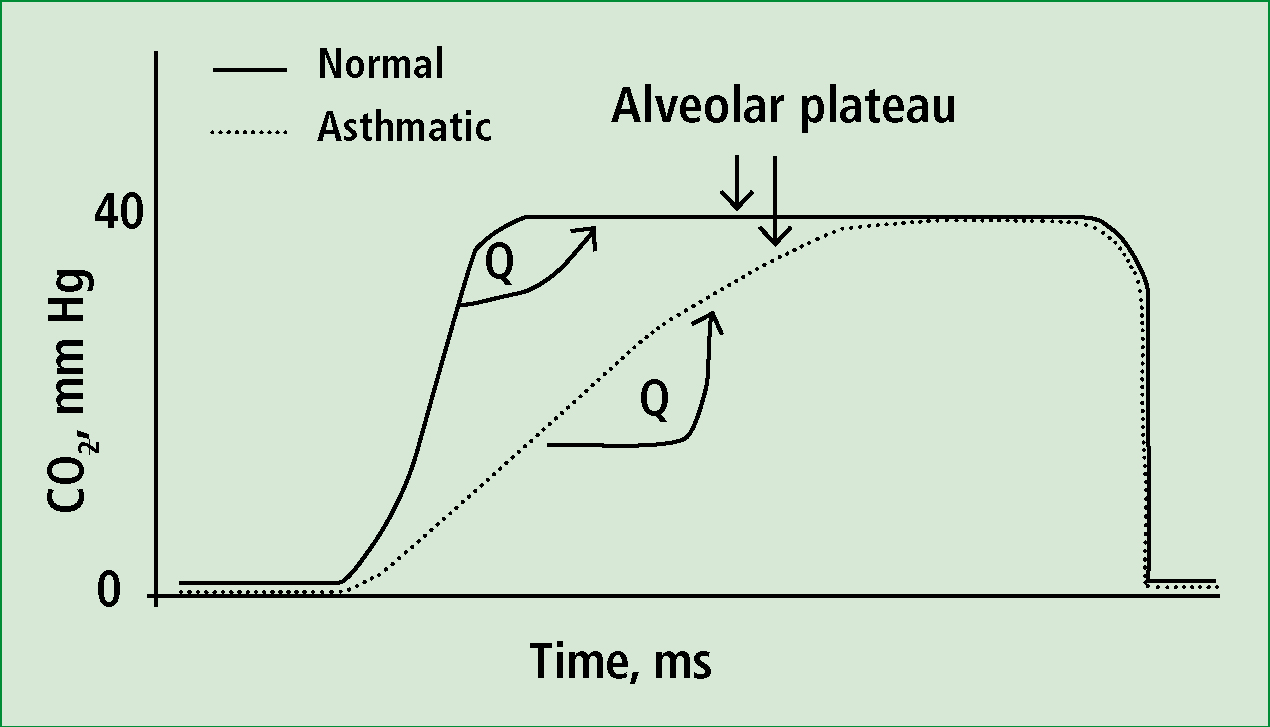
Nasal cannula-style capnography for non-intubated, self-ventilating patients has applications in assessing respiratory distress of bronchospastic nature, while also illustrating the effectiveness of interventions. Monitoring achieves this through three evaluations of the capnogram: respiratory rate, ETCO2 level and waveform shape. Respiratory rate and ETCO2 level allow an objective assessment of the severity of illness while Abramo et al (1997) described analysis of the capnogram shape as a quantitative method for evaluating degree of bronchospasm; the uneven alveolar emptying in such a disease process illicits a characteristic ‘shark-fin’ waveform, as exhalation is prolonged.
Krauss et al (2008) studied ‘shark-fin’ capnograms, reporting that ‘(waveform) changes correlate with bronchospasm, and the magnitude of the change increases with the severity of the respiratory impairment’, concluding that capnography may be used to evaluate respiratory disease state. This offers us an opportunity to assess COPD, asthma and anaphylaxis patients’ respiratory function more objectively, where an initial ‘shark-fin’ capnogram should return to a normal shape following effective intervention with nebulized bronchodilator.
Ventilation state
Respiratory rate is the single most unreliably recorded vital sign in the emergency department (Lovett 2005), yet is a vital component of many clinical presentations in all aspects of healthcare. Capnography has been examined to determine whether it can be used as a tool for accurately verify respiratory rate in an effort to avoid secondary injury from its under/overestimation; a number of studies have successfully demonstrated the benefits of capnograph-led management of respiratory rate in traumatic brain injury and major trauma, where blood gas levels are vital in outcome (Davis et al, 2003; Warner et al, 2009).
Crucially for transferability, a number of these were also conducted in the prehospital setting (Palmon et al, 1996; Helm et al, 2002; Helm et al, 2003; Davis et al, 2004; Caulfield et al, 2009) which describe the detrimental effects on outcome of unmonitored respiratory rate and endorse capnography as an adjunct to ensure accurate control. Deitch et al (2010) examined its role in reducing hypoxic events (secondary to respiratory depression) during elective sedation/ analgesia in an emergency department.
As a prospective randomized controlled trial, it ranks highly in the hierarchy of research medicine and provides strong evidence to influence clinical practice. One hundred and thirty two patients were randomly assigned to a standard (capnography) or blinded study group, reducing internal bias and increasing validity through study design; hypoxic episodes were defined as SpO2 levels of <93% and respiratory depression was defined as a loss of ETCO2 waveform for >15 seconds.
Hypoxic events were significantly less frequent in the intervention group and demonstrated statistical significance, allowing us to conclude that capnography reduces hypoxic episodes through timely intervention from earlier alerting of respiratory rate depression.
In discussing the study, the authors effectively support their results by systematically excluding confounding variables such as drug dosages, machine error and oxygen flow systems, creating greater reliability in the clinical setting. Although hospital-based, the study's essential elements of respiratory rate recording are unconditionally transferable to the prehospital arena, where the same monitoring systems can now be used.
Determining PaCO2
Some of the major issues arising from capnography use centre around its comparative accuracy to ‘true’ arterial PaCO2 levels (Ahrens and Sona, 2003) and whether the less invasive technique can comprehensively be relied upon to base clinical decisions on. This is largely attributed to the ‘ventilation/perfusion mismatch’ theory: ETCO2 and PaCO2 may differ markedly if an area of the lung is unperfused or conversely perfused but not ventilated, both common occurrences in many pathophysiologies (Ahrens and Sona, 2003).
No randomized controlled trials, systematic reviews or meta-analyses were found during the literature search, suggesting a lack of high quality, dependable research on this specific subject. Recent non-randomized trials have debated heavily: Corbo et al (2005) studied 39 patients assessing ETCO2 monitoring in determining severity of asthma, and determined a mean difference of 1 mmHg between ETCO2 and PaCO2, within the ±5 mmHg pre-specified limits of agreement.
The small convenience sample of this work leaves it open to criticism over reliability, although similar studies have described comparable results (Stein et al, 2006; Sakata et al, 2009).
Many competing articles have demonstrated incongruity (Jabre et al, 2009; Law et al, 2009; Kartal et al, 2010) although yet again, this study sampled a small number, was non-blinded and also had no exclusion criteria. A wide variety of dyspnoeic patients were included, diluting the validity of the subgroups even further (for example, only 7 asthmatic patients were studied) and significantly reducing reliability for the disease group.
| Intubation aid | Respiratory rate/pattern indicator |
| CPR indicator | ROSC/LOSC indicator |
| Role in DOD | Bronchospasm indicator |
| Hypoxic drive indicator | LVF differential Metabolic acidosis indicator |
Analysis and discussion
Current use
Capnography is currently available in two formats: adaptor for ETT and a nasal cannula style for self-ventilating patients. The overwhelming principal usage of the monitoring technique appears to be in cardiac arrest scenarios, for determining ETT position. Anecdotal evidence appears to suggest limited use outside of this setting.
‘No randomized controlled trials, systematic reviews or meta-analyses were found during the literature search, suggesting a lack of high quality, dependable research on this specific subject’
Potential use
Review of the literature around capnography exposes some appealing and original proposals for use outside of an intubation adjunct, all of which are highly reproducible and relevant to the prehospital environment; links to practice will be demonstrated.
‘The prehospital phase seems to be the most critical interval in determining the ultimate outcome after traumatic brain injury.’ (Helm et al, 2002)
This statement reiterates the importance of managing ventilation in patients sensitive to fluctuations in ETCO2 levels, rendering capnography a commanding instrument in determining patient outcome through maintenance of respiratory rates and ETCO2 levels.
This theory of respiratory observation can be extended to any haemodynamically unstable patient where scrutiny of respiratory status is so essential, providing a breath-by-breath recording of ventilatory effectiveness. Cerebrovascular events, acute respiratory disorders, seizure states, drug/ alcohol abuse and sepsis all present opportunities for closer and more effectual monitoring of breathing through capnography.
In the ‘A-B-C’ assessment protocol, SpO2 is introduced as a monitoring tool in the ‘Breathing’ phase as a measure of oxygenation. A good indicator, it is however subject to degrees of motion artefact, perfusion problems and other interferences providing erroneous readings. It should be questioned as to whether SpO2 truly reflects ‘B’; its use only provides us with a fraction of the picture. Combining this observation with capnography offers an opportunity to more effectively evaluate breathing and ventilation (where equipment is available and practical) by obtaining information about oxygenation, ventilation and perfusion to objectively assess this phase.
Potential applications in the cardiac arrest scenario extend far beyond insertion of an ETT; capnography can be used to provide feedback to optimize chest compressions, where the waveform amplitude changes with compression depth (as a reduced intensity produces a lower exhaled ETCO2). Through this method, monitoring ETCO2 during cardiac arrest may detect unrecognized compression provider fatigue (Ochoa, 1998) and is significantly more accurate than observing electrocardiograph lead oscillations, which are subject to many external artefacts.
With the updated UK Resuscitation Council (2010) guidelines placing a profound emphasis on good quality chest compressions, this creates an opening for capnography use in cardiac arrest to assess for accurate compression rate and effectiveness, allowing for feedback and subsequent increased performance if appropriate. Additionally, the technique could potentially provide a prognostic role in diagnosis of death (DOD) where specific parameters for instigation/ termination of resuscitation are assessed; initial low (<10 mmHg) ETCO2 could identify situations where resuscitation attempts are likely to be unsuccessful.
Accuracy of ETCO2 in determining PaCO2 is a key area of study, previously discussed in the review of literature. A major inconsistency still exists between researchers when evaluating whether ETCO2 is accurate enough to base significant clinical decisions upon, with many concluding that the technique is unreliable for this application when a ventilation/perfusion mismatch may exist. A middle ground must be found: arterial blood gas (ABG) measurement is impracticable in the prehospital environment, largely due to its expense and invasive nature.
An estimate of ETCO2 level and trends or changes in capnogram morphology is more important than a single reading and of greater interest to the prehospital practitioner, where interventions (i.e. nebulizers, bag-valve-mask ventilation, opiate antagonists) can be evaluated and modified accordingly.
The variance in relation to PaCO2 must be accepted as compromize when critically judging capnography as an easily used, non-invasive and timely diagnostic indicator.
Device improvements may increase reliability of ETCO2 in ventilation/ perfusion mismatch; Kasuya et al (2009) concluded that an ‘oral scoop’ (a design variation of non-invasive sidestream capnography which ‘guides’ exhaled breath toward the sampling device) improved ETCO2 performance and reduced discrepancy.
These devices are now commonly available. Such advances have recently seen nasal cannula capnography introduced in the hospital critical care setting for self-ventilating patients in level 2 High Dependency Units (HDU), where trends in capnograms and ETCO2 levels over time indicate when the more accurate ABG may be required, but removes the previously used requisite for frequent invasive and painful procedures (Grant, 2011).
Other studies around capnography include a potential differential diagnosis tool (by essentially excluding bronchospasm as the cause for the presenting respiratory distress) when assessing pulmonary oedema (commonly seen in left ventricular failure) as the alveoli are, for the most part, emptying equally and therefore produce a ‘normal’ capnogram (Warner et al, 2009).
‘With the updated UK Resuscitation Council (2010) guidelines placing a profound emphasis on good quality chest compressions, this creates an opening for capnography use in cardiac arrest’
Further potential uses comprise predicting the presence and severity of metabolic acidosis (commonly seen in DKA and sepsis) through capnography, as the compensatory tachypnoea yields a lower ETCO2. Fearon and Steele (2002) found the relationship between readings and acidotic state is both accurate and predictable, further validated in a larger study in 2006 by Agus et al.
Research is also being undertaken to examine its potential use in an early sepsis warning indicator (McGillicuddy et al, 2008), a role in predicting pulmonary embolism (through identification of a specific oscillating capnogram shape, Kim et al (2009) and as a prognostic marker in trauma surgery (Tyburski et al, 2003).
Conclusion
While its use is well established in critical care, the equipment has also found to be insufficiently used in a number of emergency departments (Delorio, 2005; Langhan and Chen, 2008) suggesting that emergency medicine lies behind in terms of using novel technology in advancing patient care. Effective and appropriate use of capnography in prehospital care has the capabilities to drive full spectrum use into the emergency department. Although an adjunct to clinical assessment, it remains a powerful tool in diagnosis and observation if used appropriately, in addition to the core essential assessment skills.
The lack of randomized controlled trials and systematic reviews into capnography require addressing. Until reliable, high quality evidence-based research exists then policy changes and further introduction into practice will be delayed. Further study into the technique and its capabilities are required. The advancing paramedic profession, with the development of informed clinicians capable of conducting research, should be in a prime position to drive the use of capnography forward.
Through creation of further prehospital studies, it may establish itself as a valuable assessment adjunct and allow us to directly affect patient intervention and outcome. Capnography is currently not mentioned in NICE (National Institute for Health and Clinical Excellence) (2004) guidelines for the management of COPD or asthma, nor in the BTS (British Thoracic Society) (2003) protocol for assessment, offering up an opportunity for prehospital research to steer ETCO2 monitoring into widespread clinical use in a variety of referral pathways, building upon the recent government White Paper ‘Liberating the NHS’ (2010) which encourages patient choice outside of the hospital and encourages empowering clinicians. This will evolve as prehospital care continues to be increasingly recognized as an essential part of a patients’ care and guidelines are produced accordingly.
